 For instance, fake tips on how to prevent or even cure Coronavirus (COVID-19) have gone viral and so have hoaxes aimed at spreading confusion. This pandemic has brought out a really clear picture of the kinds of things that tend to circulate in the misinformation ecosystem, generally, just more intensified and, obviously, with higher stakes.
For instance, fake tips on how to prevent or even cure Coronavirus (COVID-19) have gone viral and so have hoaxes aimed at spreading confusion. This pandemic has brought out a really clear picture of the kinds of things that tend to circulate in the misinformation ecosystem, generally, just more intensified and, obviously, with higher stakes.
So SKLAR Instruments has compiled here the key primary reference sources to remove any doubt on how to best manage infection control in healthcare facilities from global health authorities the Centers for Disease Control and the World Health Organization Rolling Update. Johns Hopkins University Map of USA also has a great tracker which you can also view by World or City and a further section to review Critical Trends. The Federation of American Scientists have put together a very good website with the latest global and national news. Local health authorities are very important to inform on guidelines at the local level.
MultiState COVID-19 Policy Tracker
HEALTHCARE INFECTION CONTROL PRACTICES
WHAT THE CDC HAS POSTED FOR HEALTHCARE FACILITIES
HICPAC is a federal advisory committee appointed to provide advice and guidance to DHHS and CDC regarding the practice of infection control and strategies for surveillance, prevention, and control of healthcare-associated infections, antimicrobial resistance and related events in United States healthcare settings.
CDC ENVIRONMENTAL INFECTION CONTROL GUIDELINES FOR HEALTHCARE FACILITIES

NOTE: THE CDC DOES NOT MENTION THE USE OF ALCOHOL FOR INFECTION CONTROL GUIDELINES IN RELATION TO "HEALTHCARE FACILITIES" IN RELATION TO SURFACE DISINFECTANTS. (As shown in the above screen capture from the CDC Website)
Misinformation about this in the healthcare industry seems to be a result of person to person interpretation and/or using information from a news report, that was geared to households, regarding the CDC Recommendations for “Communities, Schools and Workplaces” that states, a 70% Alcohol solution can be used as an alternative “When EPA-Approved Disinfectants are Not Available” (To reemphasize, this portion of the CDC website Recommendations is not part of the Guidelines for Healthcare Workers/Facilities).
MORE COVID-19 RESOURCES
EPA's Coronavirus Site
CDC's Coronavirus Disease 2019 Site
CDC's Cleaning and Disinfection Recommendations for COVID-19
NPIC 's COVID-19 Virus Fact sheet
How does EPA know that the products on List N work on SARS-CoV-2?
While surface disinfectant products on List N have not been tested specifically against SARS-CoV-2, the cause of COVID-19, EPA expects them to kill the virus because they:
- Demonstrate efficacy (e.g. effectiveness) against a harder-to-kill virus; or demonstrate efficacy against another type of human Coronavirus similar to SARS-CoV-2.
- All surface disinfectants on List N can be used to kill viruses on surfaces such as counters and doorknobs. Because SARS-CoV-2 is a new virus, this pathogen is not readily available for use in commercial laboratory testing to see if a certain disinfectant product is effective at killing the virus. If EPA Reg. No. 12345-12 is on List N, you can buy EPA Reg. No. 12345-12-2567 and know you’re getting an equivalent product.
70144-1 – Disinfectant (2 minute EPA listed contact time against SARS-CoV-2)
70144-2 – Disinfectant Wipes (3 minute EPA listed contact time against SARS-CoV-2)
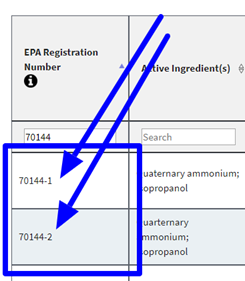
Broad spectrum anti-microbial agent effective against a wide range of pathogens and has no deleterious effects on surfaces. Completely kills infectious disease-causing micro-organisms within a few minutes. Sklar Disinfectant has the equivalent product as 70144-1 and 70144-2.
(Not for use on surgical instruments).
ALSO AVAILABLE
SKLAR Disinfectant 24 oz Spray Bottle
(2 minute EPA listed contact time against SARS-CoV-2)
10-1643 (CASE OF 6) | EPA REG. #70144-1
SKLAR DISINFECTANT SURFACE WIPES
(CASE OF 6)
EPA REG. #701442-2
(3 minute EPA listed contact time against SARS-CoV-2)
SKLAR DISINFECTANT PRODUCT INFORMATION